On December 22nd, Handelnine Global Limited DBA Navafresh announced a recall of Virgo Rheumacare Capsules due to potential lead contamination. The company recalled lot numbers CAM040 and CAL079-N. The capsules are black and purple and packaged in white plastic containers with 30 capsules in each.
The product was distributed nationwide via the Navafresh website. One adverse complaint related to this recall has been received by the company to date. Consumers are advised to report adverse reactions or quality problems to the FDA’s MedWatch Adverse Event Reporting program:
- Regular Mail or Fax: Download form www.fda.gov/MedWatch/getforms.htm OR call 1-800-332-1088 to request a reporting form, then complete and return to the address on the pre-addressed form or submit by fax to 1-800-FDA-0178.
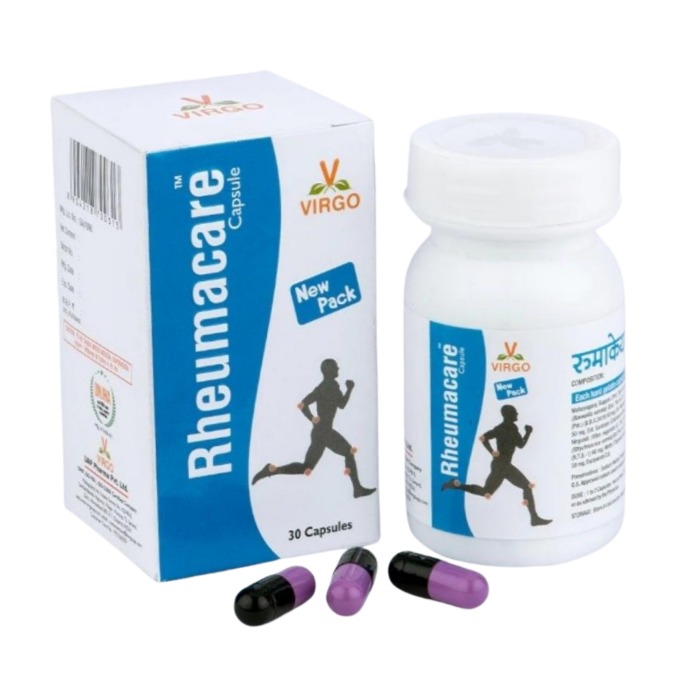 Rheumacare Capsules, Virgo
Rheumacare Capsules, Virgo
Image Source: FDA
Why Is This a Concern?
The Rheumacare Capsules are an Ayurvedic herbal supplement marketed to relieve joint pain. FDA testing found extremely high levels of lead in the product (up to 11,100 parts per million), which far exceed permissible levels. Some traditional Ayurvedic medicines may contain heavy metals such as lead, mercury, or arsenic that are not always disclosed on the label or properly controlled during manufacturing.
There is no safe level of exposure to lead. Lead can build up in the body over time, and even small amounts can cause serious health effects, including abdominal pain, nausea, anemia, nerve damage, and reproductive health complications such as miscarriage or infertility. Since this product is taken orally and may be used regularly for chronic pain, it poses a significant public health risk, particularly for pregnant people, children, and people who use herbal or traditional medicines frequently.
Ayurvedic products like Rheumacare are not FDA-approved. Consumers may not be aware of the health risks associated with unapproved Ayurvedic medicines. The FDA has warned that using products with harmful levels of heavy metals may lead to heavy metal poisoning, especially with ongoing use.
What Should You Do?
- Do not use this product or give this to your child.
- Return the product for a full refund and discard immediately.
- Call your healthcare provider if you are concerned about potential lead exposure and ask about a blood lead level test.
- Questions? Contact the company at +1 442-223-0999 or at legal@handelnine.com, Monday to Friday, 10 AM – 5 PM GMT.
Read the full Recall Announcement here.

